Data and methods
For technical notes on the state and territory summaries refer to Technical notes - state and territory summaries.
Age
Age is calculated as at the start of the episode.
Counting clients
Every client in the Alcohol and Other Drug Treatment Services National Minimum Data Set (AODTS NMDS) is assigned a statistical linkage key (SLK-581).
Clients are counted based on the number of SLK-581s in the AODTS NMDS.
National counts are based on the first time a client’s SLK-581 appears in the AODTS NMDS. All clients are counted once.
State and territory counts are based on counting the occurrence of SLK-581s for each client in the AODTS NMDS in each jurisdiction. This may result in clients being counted more than once. This is most common among clients who travel interstate for treatment. For example, clients who reside in Queanbeyan, NSW and travel to Canberra, ACT for treatment. This means that the sum of clients at the state and territory level can be greater than the national total.
This report uses both national and state and territory counts to describe trends at both national and jurisdictional levels, as well as movements between jurisdictions. For more information, refer to the supplementary table footnotes and the SLK-581 guide for use.
Data collection process
For most states and territories, the data provided for the national collection are a subset of a more detailed jurisdictional data set used for planning and policy. Figure A1 shows the processes involved in constructing the national data.
Figure A1: Alcohol and other drug treatment data collection flowchart
Drugs of concern
The Alcohol and Other Drug Treatment Services National Minimum Data Set (AODTS NMDS) contains data on drugs of concern that are coded using the ABS’s Australian Standard Classification of Drugs of Concern (ASCDC) (ABS 2011). In this report, these drugs are grouped (Table A1).
Group | ASCDC codes | Category | Includes |
|---|---|---|---|
Analgesics | 1000–1999 | Codeine |
|
| Morphine |
| |
| Buprenorphine |
| |
| Heroin |
| |
| Methadone |
| |
| Other opioids | Oxycodone, fentanyl, pethidine | |
| Other analgesics | Paracetamol | |
Sedatives and hypnotics | 2000–2999 | Alcohol | Ethanol, methanol and other alcohols |
| Benzodiazepines | Clonazepam, diazepam and temazepam | |
| Other sedatives and hypnotics | Ketamine, nitrous oxide, barbiturates and kava | |
Stimulants and hallucinogens | 3000–3999 | Amphetamines | Amphetamine, dexamphetamine and methamphetamine |
| Ecstasy (MDMA) |
| |
| Cocaine |
| |
| Nicotine |
| |
| Other stimulants and hallucinogens | Volatile nitrates, ephedra alkaloids, phenethylamines, tryptamines and caffeine | |
Cannabinoids | 7000–7199 | Cannabis |
|
Other | 4000–6999 9000–9999 | Other | Anabolic agents and selected hormones, antidepressants and antipsychotics, volatile solvents, diuretics and opioid antagonists |
Not stated | 0000–0002 | Not stated |
|
In this report, pharmaceutical drugs were grouped using 10 drug types, making up the pharmaceuticals group for the purposes of the analysis. These drugs correspond to the ASCDC codes and classifications (Table A2).
Drug category | ASCDC | ASCDC classification | Drug description |
|---|---|---|---|
Codeine | 1101 | Analgesics Organic opiate analgesics | Codeine |
Morphine | 1102 | Analgesics Organic opiate analgesics | Morphine |
Buprenorphine | 1201 | Analgesics Semisynthetic opioid analgesics | Buprenorphine |
Oxycodone | 1203 | Analgesics Semisynthetic opioid analgesics | Oxycodone |
Methadone | 1305 | Analgesics Synthetic opioid analgesics | Methadone |
Benzodiazepines | 2400–2499 | Sedatives and hypnotics Benzodiazepines | Benzodiazepines n.f.d., alprazolam, clonazepam, diazepam, flunitrazepam, lorazepam, nitrazepam, oxazepam, temazepam, benzodiazepines n.e.c. |
Steroids | 4000–4999 | Anabolic agents and selected hormones Anabolic androgenic steroids Beta2 agonists Peptide hormones, mimetics and analogues Other anabolic agents and selected hormones Not further defined | Anabolic agents and selected hormones n.f.d., anabolic androgenic steroids n.f.d., boldene, dehydroepiandrosterone, fluoxymesterone, mesterolone, methandriol, methenolone, nandrolone, oxandrolone, stanozolol, testosterone, anabolic androgenic steroids n.e.c., beta2 agonists n.f.d., eformoterol, fenoterol, salbutamol, beta2 agonists n.e.c., peptide hormones, mimetics and analogues n.f.d., chorionic gonadotrophin, corticotrophin, erythropoietin, growth hormone, insulin, peptide hormones, mimetics and analogues n.e.c., other anabolic agents and selected hormones n.f.d., sulfonylurea hypoglycaemic agents, tamoxifen, thyroxine, other anabolic agents and selected hormones n.e.c. |
Other opioids | 1100, 1199, 1200, 1299, 1300–1304, 1306–1399 | Analgesics Organic opiate analgesics Semisynthetic opioid analgesics Synthetic opioid analgesics Not further defined | Organic opiate analgesics n.f.d., organic opiate analgesics n.e.c., semisynthetic opioid analgesics n.f.d., semisynthetic opioid analgesics n.e.c., synthetic opioid analgesics n.f.d., fentanyl, fentanyl analogues, levomethadyl acetate hydrochloride, meperidine analogues, pethidine, tramadol, synthetic opioid analgesics n.e.c. |
Other analgesics | 0005, 1000, 1400–1499 | Analgesics Non-opioid analgesics Not further defined | Analgesics n.f.d., non-opioid analgesics n.f.d., acetylsalicylic acid, paracetamol, ibuprofen, non-opioid analgesics n.e.c. |
Other sedatives and hypnotics | 2000, | Sedatives and hypnotics Anaesthetics Barbiturates Gamma-hydroxybutyrate (GHB) type drugs and analogues Other sedatives and hypnotics | Sedatives and hypnotics n.f.d., anaesthetics n.f.d., ketamine, nitrous oxide, phencyclidine, propofol, anaesthetics n.e.c., barbiturates n.f.d., amylobarbitone, methylphenobarbitone, phenobarbitone, barbiturates n.e.c., GHB-type drugs and analogues n.f.d., GHB, gamma-butyrolactone, 1,4-butanediol, GHB-type drugs and analogues n.e.c., other sedatives and hypnotics n.f.d., chlormethiazole, kava lactones, zopclone, doxylamine, promethazine, zolpidem, other sedatives and hypnotics n.e.c. |
n.f.d – not further defined; n.e.c – not elsewhere classified.
Jurisdictional notes regarding principal drug of concern:
- Victoria reported comparatively high incidences of ‘Not stated drugs’ (15%) as the drug of concern. This is in part due to service providers adjusting to changes in reporting practices associated with the implementation of a new data collection system in 2019–20. In 2020–21, these drugs of concern were coded as ‘Other drugs of concern’ (14%) to realign with previous coding practices for Victoria. Victoria is working with service providers to encourage more specific reporting of drug of concern.
- In Queensland, the proportion of cannabis episodes reported as the principal drug of concern is a result of the Police Drug Diversion Program, Illicit Drugs Court Diversion Program and Drug and Alcohol Assessment Referral Program (DAAR) operating in the state.
- South Australia reports a high proportion of treatment episodes where amphetamines are the principal drug of concern due to the SA Police Drug Diversion Initiative (PDDI). In addition, adult cannabis offences are not included in the PDDI due to the SA Cannabis Expiation Notice legislation.
- In the Australian Capital Territory, removal of criminal penalties for possession of small quantities of cannabis in the ACT at the end of January 2020 reduced the number of cannabis-related diversions recorded as treatment episodes to low levels (mainly under-18s). Data collection improvements at government-operated services resulted in fewer ‘not stated’ responses in the 2022–23 collection.
Drugs of concern supplementary tables
Data for drugs of concern published in the supplementary tables may differ from results published within other tables, due to different counting methodology. Tables have been footnoted where there is different counting methodology. For example, where the principal drug of concern is coded as fentanyl (1301) and other drug of concern is coded as tramadol (1307), these drugs are within the same drug grouping (synthetic opioid analgesics) and counted only once.
Duration
Duration is calculated in whole days, and only for closed episodes.
Population rates
In this publication, crude rates were calculated using the ABS’s estimated resident population at the midpoint of the data range: that is, rates for 2022–23 data were calculated using the estimated resident population at 31 December 2022.
The COVID-19 pandemic and the resulting Australian Government closure of the international border from 20 March 2020 caused significant disruptions to the usual Australian population trends. This report uses Australian Estimated Resident Population (ERP) estimates that reflect these disruptions.
In the year July 2020 to June 2021, the overall population growth was much smaller than the years prior and, in particular, there was a relatively large decline in the population of Victoria. ABS reporting indicates these were primarily due to net-negative international migration; for further information, refer to National, state and territory population, June 2021.
Note that this change in the usual population trends may affect interpretation of statistics calculated from these ERPs. For example, rates and proportions may be greater than in previous years due to decreases in the denominator (population size) of some sub-populations.
Reason for cessation
The AODTS NMDS contains data on the reason an episode ended (reason for cessation). In this report, these reasons are grouped (Table A3), but data for the individual end reasons are available in the online supplementary tables.
A different method was used for grouping end reasons in reports released before 2014, so trend comparisons across reports should be made with caution. It is possible to compare data at the individual end reasons using the supplementary tables.
Outcome type | Reason for cessation |
|---|---|
Expected/planned completion | Treatment completed |
Ceased to participate at expiation | |
Ceased to participate by mutual agreement | |
Ended due to unplanned completion | Ceased to participate against advice |
Ceased to participate without notice | |
Ceased to participate due to non-compliance | |
Referred to another service/change in treatment mode | Change in main treatment type |
Change in delivery setting | |
Change in principal drug of concern | |
Transferred to another service provider | |
Other | Drug court or sanctioned by court diversion service |
Imprisoned (other than drug court sanctioned) | |
Died | |
Other | |
Not stated |
Remoteness area
This report uses the ABS’s Australian Statistical Geography Standard (ASGS) Edition 3 (ABS 2021) to analyse the proportion of AOD treatment agencies by remoteness area. This structure allows areas that share common characteristics of remoteness to be classified into broad geographic regions of Australia. These areas are:
- Major cities
- Inner regional
- Outer regional
- Very remote
- Remote
The remoteness structure divides each state and territory into several regions based on their relative access to services.
Examples of urban centres in each remoteness area are:
- Major cities: Canberra, Newcastle
- Inner regional: Hobart, Bendigo
- Outer regional: Cairns, Darwin
- Remote: Katherine, Mount Isa
- Very remote: Tennant Creek, Meekatharra.
For this report, the remoteness area of the agency was determined using the Statistical Area Level 2 (SA2) of the agency. Not all SA2 codes fit neatly within a single remoteness category, and a ratio is applied to reapportion each SA2 to the applicable remoteness categories. As a result, it is possible that the number of agencies in a particular remoteness category is not a whole number. After rounding, this can result in there being ‘<0.5%’ agencies in a remoteness area, due to the agency’s SA2 partially crossing into the remoteness area.
The Australian Statistical Geography Standard ASGS has replaced the Australian Standard Geographical Classification 2006 (ABS 2006), which was used in previous reports to calculate remoteness areas. Therefore, remoteness data for 2011–12 and previous years are not comparable with those for 2012–13 and subsequent years.
Service sectors
From 2008–09, agencies funded by the Department of Health and Aged Care under the Non-Government Organisation Treatment Grants Program (NGOTGP) were classified as non‑government agencies. Before this, many of these agencies were classified as government agencies. As a result, trends in service sectors of agencies should be interpreted with caution.
Source of referral – diversion
Throughout Australia, there are programs that divert people who have been apprehended or sentenced for a minor drugs offence from the criminal justice system. Many of these diversions result in clients receiving drug treatment services, who have been referred to treatment agencies as part of a drug diversion program. Since the 1980s, Australian governments have supported programs aimed at diverting from the criminal justice system people who have been apprehended or sentenced with a minor drugs offence.
In Australia, drug diversion program come in 2 main forms:
- Police diversion occurs when an offence is first detected by a law enforcement officer. It usually applies for minor use or possession offences, often relating to cannabis, and can involve the offender being cautioned, receiving a fine and/or having to attend education or assessment sessions.
- Court diversion occurs after a charge is laid. It usually applies for offences where criminal behaviour was related to drug use (for example, burglary or public order offence). Bail-based programs generally involve assessment and treatment, while pre‑ and post-sentence programs (including drug courts) tend to involve intensive treatment and are aimed at repeat offenders.
Treatment
The number of closed treatment episodes for counselling as a main treatment type has remained the most common treatment type for all clients over all collection years. All information included in the AODTS NMDS regarding clients and treatment services are based on a closed treatment episode (there is an end date which falls within the reporting period). Fluctuations over time in closed treatment episodes for particular treatment types may be influenced by coding practices, increased funding or changes in treatment policies or capacity to provide specialised alcohol and other drug treatment services, which may contribute to variation in treatment types over time.
Trends
Trend data may differ from data published in previous versions of Alcohol and other drug treatment services in Australia, due to data revisions.
From the inception of the AODTS NMDS, data have been collected only about treatment episodes provided by AOD treatment services. Data about the clients those episodes relate to have not been available at a national level. A Statistical Linkage Key-581 (SLK) was introduced into the AODTS NMDS for the 2012–13 collection to enable the number of clients receiving treatment to be counted, while continuing to ensure the privacy of these individuals receiving treatment.
An imputation strategy for the collection was developed to correct for the impact of invalid or missing SLKs on the total number of clients. This strategy takes into account several factors relating to the number of episodes per client and makes assumptions relating to spread across agencies. It also takes into consideration the likelihood that an episode with a missing SLK relates to a client that has already been counted through other episodes with a valid SLK.
To ensure an accurate representation of the AODTS client population, imputation was applied to the 2012–13, 2013–14 and 2015–16 AODTS NMDS to account for the proportion of valid SLKs being less than 95% for these years. The national rate of valid SLKs for these years was largely affected by low proportions of valid SLKs in New South Wales.
Attributing the number of clients to a set of records missing SLK
The AODTS NMDS collects information at the service record level. Service records are associated with individual clients through an SLK. There are a number of records that have missing or invalid SLK data that cannot be attributed to a client. This leads to an under‑reporting of the total number of clients using the services, because some (but not all) of the records will belong to clients who are not observed via a valid SLK.
This document describes the method of using the available data—after making several assumptions about the behaviour of the whole population—to estimate the total number of clients.
Imputation groups
Imputation groups are formed to improve the performance of the estimates. The service records were grouped according to properties that are thought to influence the behaviour of clients and the quality of SLK data, and then the imputation was performed at this imputation group level.
Possible properties used to develop groups include location, provider size (measured by number of service records) and service type. The data are also grouped according to any subpopulations that are going to be reported upon, such as jurisdiction.
The final imputation groups were formed by balancing the often-competing priorities of having homogenous groups and the need to have groups large enough to ensure that the imputation is robust.
Assumptions and approximations
Assumption 1: randomness and independence
This imputation method assumes that whichever service provider a client attends for each incidence of service is random and independent of any other incidents of service the client may have. It is further assumed that the validity or otherwise of the SLK recorded on each service record is random, and independent of both the client and the service provider with which the record is associated.
Assumption 2: distribution of the number of service records per client
This method also assumes that the distribution of the number of records per client for all clients is similar to that observed using the subset of records with valid SLKs.
Approximation 1: no client has more than 10 service records
This imputation method uses the approximation that no client has more than 10 service records.
In order to implement this approximation, any clients observed to have more than 10 service records were treated as if they had only 10, and the proportion of clients with 10 service records calculated accordingly.
Notation
The definition of the notation used in this document is as follows:
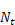 : the (unknown) total number of clients
: the (unknown) total number of clients
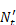 : the imputed total number of clients
: the imputed total number of clients
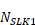 : the number of clients observed using the records with a valid SLK
: the number of clients observed using the records with a valid SLK
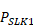 : the proportion of clients with at least 1 service record with a valid SLK
: the proportion of clients with at least 1 service record with a valid SLK
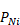 : the (unknown) proportion of clients with
: the (unknown) proportion of clients with  service records
service records
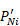 : the imputed proportion of clients with
: the imputed proportion of clients with  service records
service records
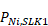 : the proportion of clients with
: the proportion of clients with  service records as observed using records with valid SLKs
service records as observed using records with valid SLKs
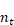 : the total number of service records
: the total number of service records
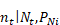 : the number of service records given the total number of clients and the proportions of clients with
: the number of service records given the total number of clients and the proportions of clients with  service records,
service records,  = 1,2, ... 10
= 1,2, ... 10
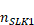 : the number of service records with a valid SLK
: the number of service records with a valid SLK
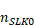 : the number of service records with an invalid SLK
: the number of service records with an invalid SLK
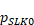 : the proportion of service records with an invalid SLK.
: the proportion of service records with an invalid SLK.
Methodology
Given Assumption 1 and Approximation 1, the proportion of clients who have at least 1 service record with a valid SLK is:
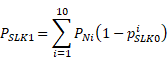
Now:
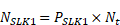
so it follows that the total number of clients is:
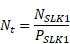
To resolve this equation for 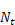 the values of the
the values of the 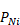 is required. These are unknown, given it is not possible to observe the whole population due to the records with invalid SLK values. This method imputes the unknown
is required. These are unknown, given it is not possible to observe the whole population due to the records with invalid SLK values. This method imputes the unknown 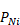 using numerical methods, then uses these values to impute
using numerical methods, then uses these values to impute 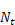 .
.
The process starts with the distribution of number of records per client that were observed using the records with valid SLKs (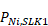 ). These values are then adjusted so that the following conditions are met.
). These values are then adjusted so that the following conditions are met.
Constraint 1
The sum of the imputed proportions is equal to 1. That is:
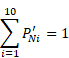
Constraint 2
The imputed proportion of clients with 1 service record is less than or equal to the observed equivalent proportion among clients with records with valid SLKs. That is:
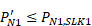
This constraint is used because some of the clients observed to have only 1 record will, in fact, have additional records with invalid SLKs. It is unlikely that the true proportion of clients with 1 service record is higher than that observed using records with valid SLKs.
Constraint 3
The total number of service records that the imputed total number of clients and the imputed distribution of records per client imply is equal to the observed number of service records.
That is:
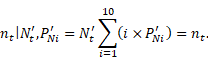
This constraint is used to ensure that the imputed values are consistent with the observed number of records.
Penalty function
Under Assumption 2 we want to limit how much the imputed proportions differ from the proportions observed via the records with valid SLK data. To achieve this we use a penalty function that increases as the distance between the imputed and observed proportions increases. This function is defined to be:
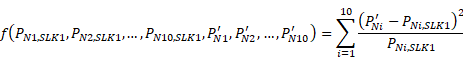
Using numerical methods, the 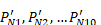 are chosen such that the penalty function is minimised, subject to the 3 constraints.
are chosen such that the penalty function is minimised, subject to the 3 constraints.
The final step is to use the imputed proportions to calculate the imputed total number of clients:
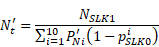
The resulting number is then rounded to the nearest integer.
Discussion
This imputation technique uses available information to impute the total number of clients. The methodology takes into account the proportion of records with invalid SLK data and the distribution of the number of service records per client, as observed via the records with valid SLK data. It is apparent that the assumptions made do not hold for every client or service record. It is reasonable to expect that a client’s attendance at a service provider will be affected by location and any prior contact they had with a provider. It should also be noted that some service providers failed to collect SLK for any service record during the reference period.
Despite the known cases where Assumption 1 does not hold, it is reasonable to hope that, across the population as a whole, the assumption is a reasonable representation of the populations of clients and service records.
It is believed that the impact of Approximation 1 will be small because, given Assumption 1, the chance that a client with more than 10 service records is not observed via a record with a valid SLK is extremely small. The chance diminishes as the proportion of records with an invalid SLK decreases and across jurisdictions the highest proportion observed is about 0.3. It should also be noted that the largest proportion of clients with 10 or more service records observed in the data at the jurisdiction level was only 0.007.
There are many different penalty functions that could be used in this imputation. The function used was chosen because, compared with the other penalty functions investigated, it produced imputed proportions that were generally as close or closer to the observed proportions. It also most consistently resulted in a distribution that was similar in shape to the observed distribution of the number of records per client.
Historical data element changes
Details on historical data element changes are found in Appendix A of the AODTS NMDS Data Collection Manual 2022–23 [PDF 735kB].
ABS (Australian Bureau of Statistics) 2006. Statistical geography: volume 1—Australian Standard Geographical Classification (ASGC). ABS cat. no. 1216.0. Canberra: ABS.
ABS 2011. Australian Standard Classification of Drugs of Concern, 2011. ABS cat. no. 1248.0. Canberra: ABS.
ABS 2016. Estimates of Aboriginal and Torres Strait Islander Australians, July 2016. ABS cat. no.1270.0.55.005. Canberra: ABS.
ABS 2021. Australian Statistical Geography Standard Edition 3. Canberra: ABS.