Medicines for cardiovascular disease
Page highlights:
Supply of cardiovascular medicines
- Almost 116 million Pharmaceutical Benefits Scheme prescriptions for cardiovascular medicines were supplied to the Australian community in 2021–22.
Use of cardiovascular medicines
- In a 2018–18 survey, 79% of the estimated 1.2 million Australian adults living with heart, stroke or vascular disease had used a cardiovascular system medicine in the last 2 weeks.
Cardiovascular medicines are key elements in preventing and treating cardiovascular disease (CVD) and its risk factors. They are most commonly used to help control levels of blood pressure and blood lipids, and to regulate heartbeat.
Cardiovascular medicines
Blood pressure lowering medicines – treat high blood pressure. They include:
- Antihypertensives – suppress signals that make the heart beat harder, or open and relax peripheral arteries.
- Diuretics – increase urination, helping rid the body of water and salt and thus reduce blood volume
- Beta-blockers – suppress signals that cause the heart to beat fast and hard
- Calcium channel blockers – block a conduction pathway in the heart, reducing the force of contraction and widening blood vessels
- Renin-angiotensin system agents – block effects of the renin-angiotensin system, a hormone system that regulates blood pressure and the volume of fluids in the body. The group includes ACE inhibitors (plain and in combinations), angiotensin II receptor blockers (plain and in combinations), and other agents acting on the renin-angiotensin system.
Lipid-modifying medicines – control blood lipid levels. Statins, resin binders, nicotinic acid, fibrates and probucol reduce blood LDL cholesterol (‘bad’ cholesterol), possibly increase HDL cholesterol (‘good’ cholesterol), and lower blood triglycerides.
Antithrombotic medicines – prevent or dissolve blood clots, reducing the risk of heart attack, or further strokes among patients with a history of ischaemic stroke.
Cardiac therapy – includes cardiac glycosides, antiarrhythmics and cardiac stimulants. They regulate heart rhythm, and treat angina and heart failure. Also includes vasodilators which open the main blood vessels of the body, as well as other cardiac preparations.
Peripheral vasodilators – open blood vessels in outer parts of the body, such as the arms and legs, making it easier for the heart to pump blood.
Vasoprotectives – relieve or prevent conditions of the blood vessels.
Supply of cardiovascular medicines
A wide range of subsidised cardiovascular medicines are made available to the Australian community through the Pharmaceutical Benefits Scheme (PBS), and through other arrangements where appropriate.
 Almost 116 million PBS prescriptions for cardiovascular medicines were dispensed to the Australian community in 2021–22. These comprised 37% of total PBS prescriptions (Department of Health and Aged Care 2022).
Almost 116 million PBS prescriptions for cardiovascular medicines were dispensed to the Australian community in 2021–22. These comprised 37% of total PBS prescriptions (Department of Health and Aged Care 2022).
- 68% of these medicines (79 million) were subsidised by the PBS, with the remainder (37 million) priced below the co-payment level.
- Rosuvastatin (15.2 million) and atorvastatin (11.8 million), both lipid-modifying medicines – and perindopril (6.9 million), a blood pressure lowering medicine – were among the most commonly supplied PBS medicines in Australia in 2021–22.
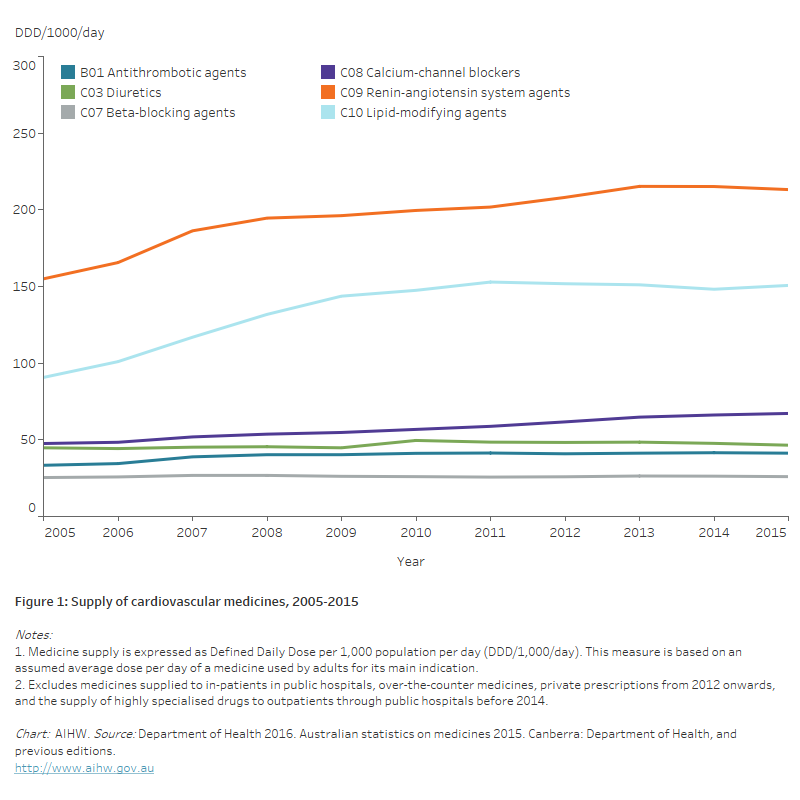
- The supply of lipid-modifying agents increased by 66% between 2005 and 2015, while calcium channel blockers and renin-angiotensin system agents – both classes of blood pressure lowering medicines – increased by 41 and 38%, and antithrombotic medicines by 24% (Figure 1).
Figure 1: Supply of cardiovascular medicines, 2005–2015
The line chart shows that between 2005 and 2015, the supply of renin-angiotensin system agents increased from 155.0 to 213.2 DDD/1,000/day, lipid-modifying agents increased from 90.7 to 150.7 DDD/1,000/day while the supply of calcium-channel blockers increased from 47.6 to 67.2 DDD/1,000/day. The supply of beta-blocking agents, antithrombotic agents and diuretics remained largely unchanged throughout the period.
Medicine supply to Australians in remote communities
Under the Remote Area Aboriginal Health Services (RAAHS) program, established under section 100 of the National Health Act 1953, any person attending an approved RAAHS can receive eligible PBS medicines without the need for a PBS prescription and without cost.
These arrangements seek to address barriers experienced by people living in remote areas of Australia, which may have limited access to a GP or a community pharmacy, in accessing essential medicines through the PBS.
- 1.7 million PBS items at a cost of $45.6 million dollars were supplied to participating Aboriginal Health Services in 2021–22 (Department of Health and Aged Care 2023).
Use of cardiovascular medicines
 More than three-quarters (79%) of the estimated 1.2 million Australian adults aged 18 and over who were living with heart, stroke or vascular disease in 2017–18 used a cardiovascular system medicine in the 2 weeks prior to survey (AIHW analysis of ABS 2019).
More than three-quarters (79%) of the estimated 1.2 million Australian adults aged 18 and over who were living with heart, stroke or vascular disease in 2017–18 used a cardiovascular system medicine in the 2 weeks prior to survey (AIHW analysis of ABS 2019).
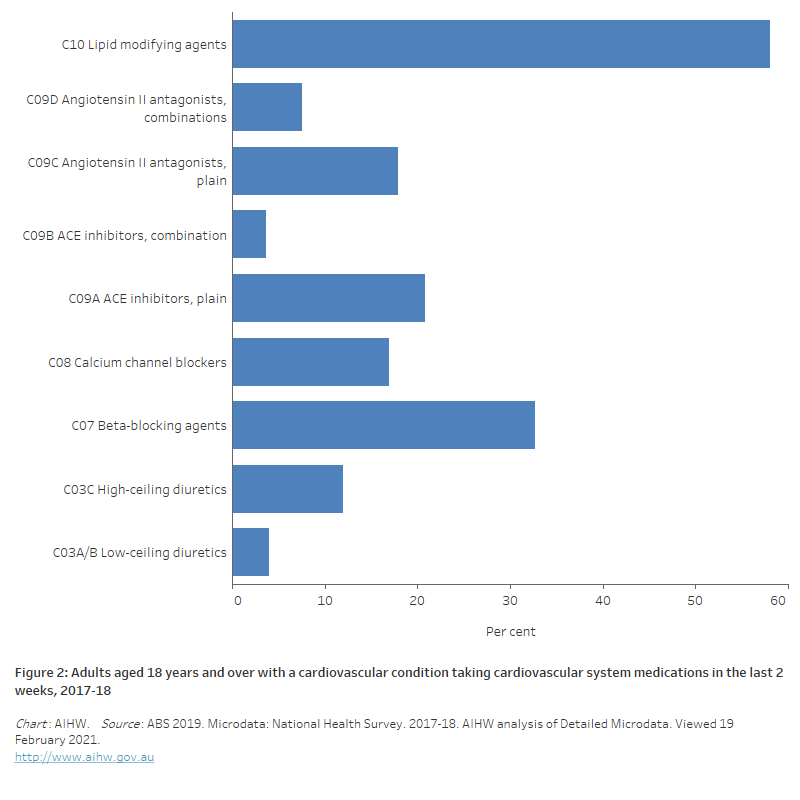
- The most common medications used were blood pressure-lowering agents—including beta-blocking agents (33%), ACE inhibitors, plain (25%), angiotensin II antagonists, plain (18%) and calcium channel blockers (17%)—and lipid-modifying agents (58%) (Figure 2). These patterns largely reflect GP prescription and PBS supply data.
- There was no statistically significant difference in the proportion of men (82%) and women (76%) using cardiovascular system medications for their condition in the previous 2 weeks.
- Use was more common in older people with heart, stroke and vascular disease, with around 22% of persons aged 18–44 years, 79% of persons aged 55–64 years, and 90% of persons aged 75 and over using a cardiovascular system medication in the previous 2 weeks.
Two-thirds (66%) of a study population of PBS concessional beneficiaries aged 65 and over used more than one class of cardiovascular medicine to manage their conditions in 2014–15. One-quarter (23%) received blood pressure lowering, lipid-modifying and antithrombotic medicines (AIHW 2017).
Figure 2: Adults aged 18 and over with a cardiovascular condition taking cardiovascular system medications in the last 2 weeks, 2017–18
The horizontal bar chart shows the most commonly used cardiovascular medications used by adults in the 2 weeks prior to survey in 2017–18 were lipid modifying agents (58.1%) followed by beta-blocking agents (32.7%) and ACE inhibitors, plain (20.8%).
ABS (2019) Microdata: National Health Survey, 2017–18. AIHW analysis of Detailed Microdata. Viewed 19 February 2021.
AIHW (2017) Medicines for cardiovascular disease. Cat. no. CVD 80. Canberra: AIHW.
Department of Health and Aged Care (2022) PBS expenditure and prescriptions report 1 July 2021 to 30 June 2022, Department of Health and Aged Care, Australian Government, accessed 1 March 2023.
Department of Health and Aged Care (2023) S100 Remote Area Aboriginal Health Services (RAAHS) Program Information Sheet, accessed 1 March 2023.